Widely-Used Diabetes Drug Recalled Due To High Levels Of Cancer-Causing Agent
By Scottie Andrew, CNN
(CNN) -- A widely-used diabetes drug has been recalled after manufacturers found it contained unacceptably high levels of a cancer-causing contaminant.
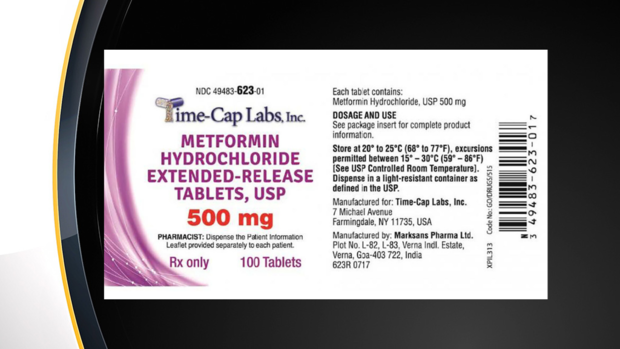
Indian pharmaceutical company Marksans Pharma Limited is recalling metformin hydrochloride extended-release tablets because their levels of NDMA, a "probable human carcinogen," were higher than the acceptable daily intake limit of 96 nanograms per day, according to a recall published this week by the U.S. Food and Drug Administration (FDA).
Metformin tablets are used to treat Type 2 diabetes and are designed to lower glucose levels.
The recall applies to metformin tablets between 500 mg and 750 mg, sold under the brand name Time-Cap Labs, Inc.
The recall expands an earlier recall of the same product from this summer. But it's just one of several metformin products that have been found to contain NDMA in the last year. Seven other pharmaceutical companies have issued recalls for metformin hydrochloride extended-release tablets due to their carcinogenic contents.
The FDA is still investigating where NDMA comes from and how it ends up in metformin products.
Most levels found in medications are generally low and fall within the FDA's accepted daily intake, but recently recalled medications exceed that. Marksans Pharma Limited, India, however, did not reveal how much NDMA its recalled products contained.
The recall applies to the following products, which can be identified by their National Drug Code numbers listed below (National Drug Codes can be used to search and identify products online through the FDA). The tablets are either embossed with 101 or 102 on one side and are plain on the other.
Metformin Hydrochloride Extended-Release Tablets, USP 500mg:
- 90 counts: 49483-623-09
- 100 counts: 49483-623-01
- 500 counts: 49483-623-50
- 1000 counts: 49483-623-10
Metformin Hydrochloride Extended-Release Tablets, USP 750mg:
- 100 counts: 49483-624-01
For much more information on this recall, visit the FDA's website here.
The-CNN-Wire
™ & © 2020 Cable News Network, Inc., a WarnerMedia Company. All rights reserved.